In this post, I discuss the composition of water that we use to brew coffee. If you are new to these discussions, I strongly recommend that you first read this previous post about brew water.
Barista Hustle recently released a very clever Excel calculator to determine the amount of mineral concentrates needed to craft brew water recipes starting from soft tap water instead of distilled water.
I thought this was a great idea, and decided to make a similar tool for those like me who prefer to use a single concentrate. While I was at it, I made it in a way that allows you to use more minerals on top of epsom salt and baking soda, which allows to control the concentration of magnesium, calcium, sodium, sulfate and bicarbonate ions individually, instead of just hardness and total alkalinity.
We are only beginning to understand the effects of magnesium, calcium and sodium ions on how fast different chemical compounds extract; this is discussed a little in the Barista Hustle water course, but so far I have not seen much more information about this elsewhere. However, I have never seen much discussion on the effects of sulfate ions on the resulting coffee taste or composition. This new water crafting tool could allow us to experiment with it while keeping everything else fixed.
The uses of this tool go even further; if you have reverse osmosis water with non-zero mineral composition, you can adapt your concentrate to still get the proper brew water composition. You could even use it to craft custom brew water starting from soft water bottles.
One thing this tool won’t allow you to do is create brew water that is lower than your tap, reverse osmosis, or water bottles in either total alkalinity, hardness, or individual ionic concentrations. This is because doing so by adding minerals is just not possible (maybe that would be possible with reactive compounds, but let’s not go there).
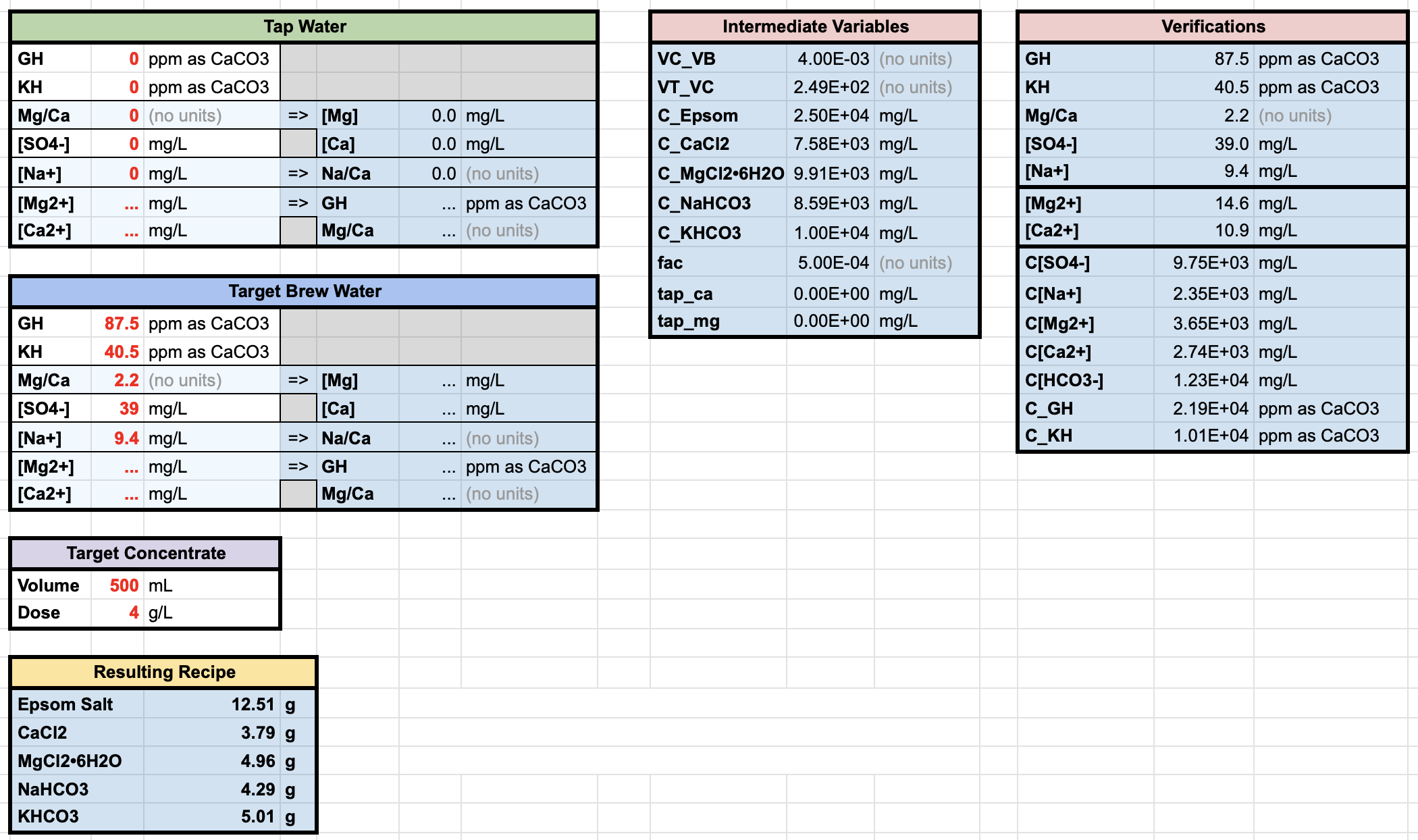
The tool I built is a Google Sheet; if you are new to Google Sheets, keep in mind that you won’t be able to modify it before you create your own copy, with File/Make a Copy. Asking me for edit permissions won’t work; doing so would modify the sheet for every other user. You can find the tool here.
Make sure to read the header instructions of the sheet. There are a few different versions of the calculator for those who don’t have access to all minerals.
I thought this was also a good moment to release publicly two of my previous Patreon-only videos related to brew water. In the first one, I filmed myself crafting a single batch of Rao/Perger brew water concentrate; you can find more explanations about the required material here;
In this second video, I use the concentrate to prepare a 4L container of brew water, starting with distilled water; you can find some more information about it here;
[Edit Nov 27, 2019: David Seng just let me know that he also built a water crafter page on his website. You should definitely check it out, as it seems very helpful and even includes the Langelier saturation index for scale and corrosion !].
I’m hoping this will help you brew better coffee, hopefully with a little less hassle for those of you lucky enough to live in a soft water area !

If I have 10ppm eg in RO and in tap water I have 140 ppm and 180 hardness and 130 alkalinity in GH do I put 12.857 and alkalinity 9.286 for Rao /Perger water? Thanks
LikeLike
Yes exactly. Your RO diluted your tap water at 10 ppm / 140 ppm so your input water has 10/140×180 = 12.9 ppm hardness and 10/140×130 = 9.3 ppm alkalinity.
LikeLike
You are very precise with everything in the mixing videos. Do you measure the water in a new 4 L QUÉBEC-O bottle before you use it? It is always more in it than 4 L.
LikeLike
I don’t have a good scale for 4L measurements. I strongly suspected that they overfilled them, it might be the biggest source of uncertainty in my brew water preparation; but it’s over a larger volume of liquid so it’s probably not too bad. Have you ever measured how much they overfill them ? Now I have a batch of Compliments Distilled water, and I think they also over-fill those.
LikeLike
All products are a bit overfilled (water, sugar, flour, etc.). Unfortunately the +extra can be different (filling systems / manufacturing sites / maintenances). The only goal companies have is to put more in it so they are save.
LikeLike
Hi Jonathan,
One simple (maybe silly) question. How do you measure your tap water? Doy ou have to measure all?
– Gh and Kh, I guess with an aquarium kit.
– Mg/Ca. I see that there are other aquarium kits that do it.
– And the rest? SO4, Na, Mg, Ca….
Thanks and regards!
LikeLike
It’s often useful to Google for your city’s latest water report. I would verify it against aquarium titration tests for GH & KH to be sure your end of the tap isn’t different, otherwise you can send a sample of your tap water to a water quality lab that can measure all of that. Most tap water has zero Mg so it’s often accurate to approximate that all GH is made of Calcium. For RO water purposes, the tap water composition is so diluted that any inaccuracies in your tap water composition are also reduced, making it less important to be very precise in determining your tap water composition.
LikeLike
Ummm… Difficult… Here the origin of the tap water changes quickly and there are no water reports in such detail.
I’ll check if I can make the test by myself. Anyway, I have free distilled water.
Thanks Jonathan!
LikeLike
Hey, you can use aquarium titration kits for GH and KH. I’d recommend measuring KH most importantly, then GH if you can, those are the most useful ones.
LikeLike
Hey Jonathan, I have a question about the coffee water spreadsheet. For the first tab recipe, for distilled water, for MgCl2•6H2O , can I substitute half the 4.96g specified with MgCl2 (2.48g)? The reason I ask is because 1) I only have MgCl2 at my disposal and 2) in your post (https://coffeeadastra.com/2018/12/16/water-for-coffee-extraction/) you specify 2g MgCl2•6H2O (hexahydrate) or 1 g anhydrous MgCl2 as part of the Rao/Perger recipe. However, I did not know if this 2:1 substitution can be applied to other similar recipes due to the chemistry rules behind it which I fully have not digested yet. Practically, I’ve made both recipes and the 500g recipe (water only) did not taste good with the substitution, but the 200g one (water only) as is on your post does taste good. Thanks!
LikeLike
Hey, yes that should work ! I never noticed anhydrous MgCl2 has exactly half the molar mass of the hexahydrate form, that’s funny. I have no idea why your 200 mL concentrate version tasted different from the 500 mL concentrate, that’s very weird.
LikeLike
Hi Jonathan, my local water report seems too vague to be helpful. It provides a range and maximum values for calcium etc. any suggestions for using this information with your calculator? Thanks Michael ihttps://www.kentwa.gov/home/showdocument?id=12230
LikeLike
I’d recommend getting an aquarium titration kit for GH & KH at a local aquarium store. This will get you the total hardness and alkalinity.
LikeLike
Hi Jonathan, I got the test kit, and my GH ppm is 53.7 and KH ppm is 17.9. Do I leave all the other entries zero?
Also, am I adding the resulting suggested mineral amounts to 200ml like the original concentrate recipe or 500ml?
Thanks Michael (moderately confused)
LikeLike
Hi Michael, I see that you commented twice – your initial comment had not appeared yet because I did not have time to approve it yet (anti spam measures). You would leave Mg/Ca to zero normally, because Mg is very rare in any tap water. Unless you know your Na+ or SO4- concentrations, you can leave them to zero too; they are usually small. So the answer is yes, you can leave everything else in the “Tap Water” section to zero. You can decide whether you want to use a 500 mL or 200 mL concentrate; just enter whatever volume of concentrate you wish to have in the cell called “Target Concentrate: Volume”.
LikeLike
Thanks for this post and the work you’ve put into the spreadsheets.
A basic question that I may have missed the answer to, if I am creating a concentrate to add to soft water, do I create the concentrate (using the recipe from the spreadsheet) with distilled water or the tap water?
LikeLike
Hello, I have been using the tool for years now. How should I set the tool if I want to bypass the concentrate. I would like to put the ingredients directly in a 5L jug but I cannot do the math.
LikeLike
Hi, set the concentrate to be used with the recipe “1000g/L” instead of “4g/L”. This will give you the recipe for the whole bottle of water instead (if you set your concentrate volume to 5L).
LikeLike